
One of the most common subtypes of non-small cell lung cancer (NSCLC) is lung adenocarcinoma, which is infamous for being misleading. The glandular cells that are normally seen in the outer region of the lungs are the source of this malignancy. Many cases remain asymptomatic in their early stages, while some people may present with symptoms such as a chronic cough or chest pain. Its discovery is made more difficult by its diverse forms, which include solid, acinar, and papillary development patterns. A patient may, for example, have a tumor with a solid predominant pattern mixed with small micropapillary or acinar patterns. Its heterogeneity determines not only how difficult it is to diagnose but also how best to treat it. Its heterogeneity, therefore, poses major obstacles to precise detection and prompt intervention, both phenotypically and molecularly.
There are numerous repercussions when lung adenocarcinoma is misdiagnosed or remains undiscovered:
Over-treatment and under-treatment: Patients are protected from over-treatment and under-treatment with a proper diagnosis. The term “over-treatment” describes the giving of needless medical care that has no positive impact on a patient’s health. Contrarily, under-treatment refers to not obtaining adequate care or getting it too late, both of which increase the risk of illness development or associated problems.
Economic impact: Financially speaking, misdiagnoses raise medical expenses. Longer hospital stays and redundant testing strain healthcare systems and place patients and their families in unnecessary financial hardship.
Psychological burden: Additionally, patients and their loved ones experience an emotional toll. The psychological upheaval that follows a cancer diagnosis might be exacerbated by discovering that one’s illness was misdiagnosed. This can breed mistrust in medical institutions.
This means that getting a precise diagnosis of lung adenocarcinoma as soon as possible is not only a medical goal but also a multifaceted necessity for patients’ overall health and the effectiveness of our healthcare systems.
Lung cancer is diagnosed using a multi-step procedure in the conventional method. Imaging tests like CT or X-rays are the first step in the process. It is usually advised to have a biopsy if an anomaly is discovered. During a biopsy, a tiny sample of lung tissue is removed, and pathologists analyze it under a microscope. The kind, grade, and stage of the tumor are ascertained by this microscopic analysis. This approach has been the gold standard for many years, but because of the complexity and range of symptoms of lung adenocarcinoma, it takes a long time and is prone to human error.
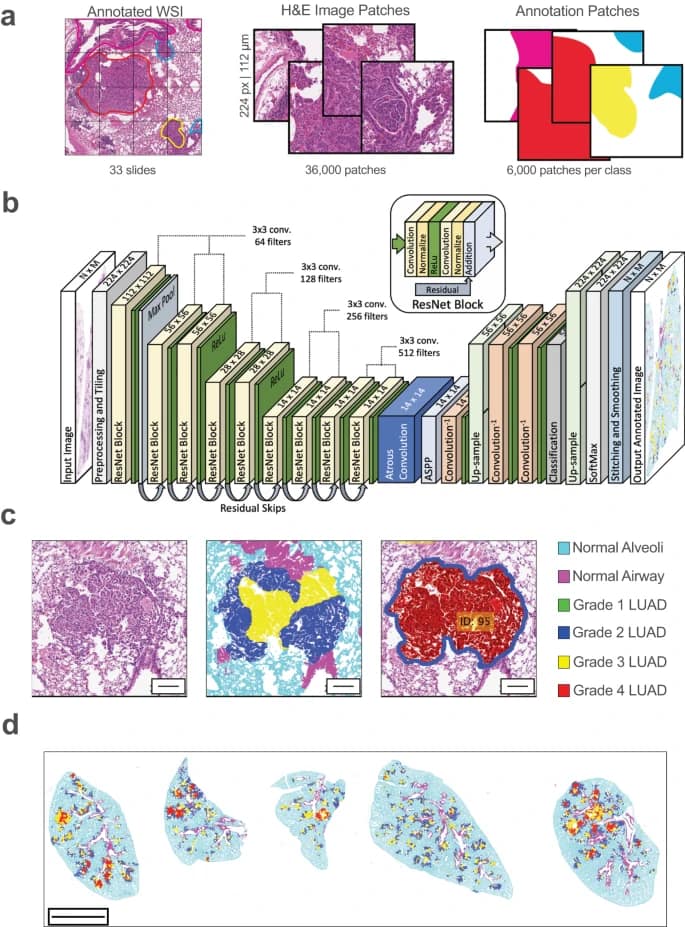
GLASS-AI, a state-of-the-art machine learning solution, is taking on this diagnostic problem and aims to enhance and optimize the diagnostic procedure. GLASS-AI, in contrast to conventional techniques, uses artificial intelligence to assess Whole Slide Images (WSIs), which are high-definition pictures of pathology slides. This novel method combines pathology and machine learning to get precise insights from these images.
At the heart of GLASS-AI is the atrous convolution layer – a special type of layer in deep learning that utilizes filters to handle different resolutions within an image. This allows for a nuanced and detailed image analysis, perfect for identifying subtle differences and patterns in complex medical slides. GLASS-AI operates as a diagnostic solution, utilizing AI to pinpoint and categorize the different stages and types of tumors, while potentially also acting as a predictive tool by identifying precancerous lesions or early-stage tumors that might escape the human eye.
What gives GLASS-AI its edge over conventional models is its sheer efficiency and accuracy:
- Speed and Efficiency: In under 75 minutes, GLASS-AI processed all 10 WSIs, which are high-resolution pictures of pathology slides. To put this into context, each slide required about 4.5 hours of labor from human raters. This amazing reduction in analysis time is essential for prompt diagnosis and medical intervention.
- Accuracy: The technology has an astounding 88% accuracy rate. Regarding tumor recognition, GLASS-AI detected tumors within 1932 of the 1958 manually segmented (specifically indicated) malignancies in the 10 WSIs. That’s not all, though. GLASS-AI identified 31% more tumor area on average when compared to human raters. This higher detection rate—which is particularly noteworthy for Grade 1 cancers—indicates its improved capacity to identify tiny tumors that the human eye could overlook.
- Reliability: Upon evaluating its alignment with human specialists in tumor grading, GLASS-AI demonstrated a Cohen’s kappa of 0.782 (95%CI: 0.759–0.806). To put it simply, a statistic called Cohen’s kappa is used to assess inter-rater agreement for categorical items. A result of 0.782 indicates a significant degree of consistency between GLASS-AI’s assessments and those of human experts; values closer to 1 indicate full agreement.
As with any breakthrough, GLASS-AI too faces its own set of challenges when transitioning from research labs to clinics and laboratories across the world:
- Model flexibility:
- Challenge: Unexpected variations may arise when transitioning from a controlled study setting to diverse real-world contexts. Within the study, GLASS-AI was tuned for a certain subset of WSIs. So, the question is, how would it function with samples from different demographics or staining methods?
- Solution: Conducting global validation research can help with this issue. Through the collection and assessment of WSIs from a wide range of populations and the use of diverse sample preparation methods, we can guarantee the model’s flexibility and resilience in a variety of contexts.
- Infrastructure and technology:
- Challenge: The availability of high-quality WSIs is a major prerequisite for GLASS-AI effectiveness. It’s possible that many low-resource environments lack the sophisticated machinery needed to create these images or the technological infrastructure needed to process them.
- Solution: A remedy may be provided by integrating GLASS-AI into cloud-based technologies (digital infrastructures offering remote data access and storage over the internet, eliminating the need for local hardware or software). In order to maximize shared technology resources, health centers with limited local computational capacity can nevertheless obtain diagnostic results by uploading their WSIs to these platforms. This allows for broader access.
- User expertise:
- Challenge: Even though GLASS-AI promises better diagnostics, understanding its outputs and complexities is still necessary. Medical professionals may face a significant learning curve in areas where pathology is only starting its digital transformation.
- Solution: Enabling focused training programs can remove this obstacle. Healthcare workers can be prepared to fully utilize the system by participating in interactive online courses, group seminars, or practical simulations, which will guarantee consistent application and interpretation guidelines.
Realizing the wider ramifications is crucial once the problems have been discovered and solutions prepared. Closing the gap between high-tech diagnostics and environments with few resources could spur a global change in healthcare toward more fair treatment. We may make progress toward global health justice by guaranteeing that GLASS-AI, a cutting-edge diagnostic tool, is understandable, accessible, and applicable in a variety of contexts. Thus, the transformational potential of AI is grounded in concrete, life-saving results, and everyone will receive a prompt and correct diagnosis, regardless of geographic or economic obstacles.
Do you see the potential for a global shift in healthcare? Share your thoughts.
Source Article: https://www.nature.com/articles/s41698-023-00419-3#Sec2
Disclaimer: Please note that the opinions, content, and analysis in my posts are entirely my own and do not reflect the views of any current or past employers or institutional affiliations. These posts, based solely on publicly available information, are for informational purposes and should not be taken as professional advice. All insights and conclusions are my viewpoints and should not be considered representative of any organizations I am or have been associated with. This content is not endorsed by, nor does it represent the stance of any affiliated entity.





